Lost and Found

With a sample of a cyanobacterium collected more than 25 years ago from the remote Pacific island of Pohnpei, a multidisciplinary team of NC State University scientists are working together to figure out how, and why, the bacterium makes a novel anticancer compound called tolyporphin.
In 1992, University of Hawaii researchers reported that a strain of Tolypothrix nodosa — its name means “knobbly hairy ball of yarn” — produced a pigment called tolyporphin A. They also reported that the pigment showed cancer-fighting potential in the lab.

Since then, scientists haven’t learned much about the strain. But Dr. Eric Miller, of NC State’s Department of Plant and Microbial Biology, and Dr. Jonathan Lindsey, of the Department of Chemistry, are changing that.
Chemistry Meets Microbiology
Using the advanced scientific tools and techniques of their disciplines – including mass spectrometry, scanning electron microscopy and high-throughput genome and metagenome sequencing – they are learning more about the strain’s unusual chemistry and biology. That could be key, Miller says, for finding ways that the compound, or its derivatives, might be put to use in medicine or other applications.
The science journal Applied and Environmental Microbiology (AEM) published a paper on Miller and Lindsey’s research in its October 2017 edition. Other authors were NC State research assistant Rebecca Hughes, graduate students Yunlong Zhang and Ran Zhang and Dr. Philip Williams, a chemist from the University of Hawaii Cancer Center.

The Backstory
In person, Miller sketches an intriguing backstory: HT-58-2 was collected in Nan Madol, once a royal city made of artificial islets surrounded by narrow stretches of water on the Micronesian island of Pohnpei. UNESCO recently designated the archaeological remains of that city a World Heritage Site.
The University of Hawaii added the sample to a collection of hundreds of kinds of cyanobacteria, then known as blue-green algae. For years, the culture had been thought lost. However, Williams recovered the culture and has been collaborating with Miller and Lindsey to learn more about it.
What Is It, Exactly?
In terms of science, what the researchers have learned so far about the strain is remarkable. When they analyzed the cyanobacterium’s ribosomal RNA, they noted that it looks more like cyanobacteria of the genus Brasilonema than it does Tolypothrix.
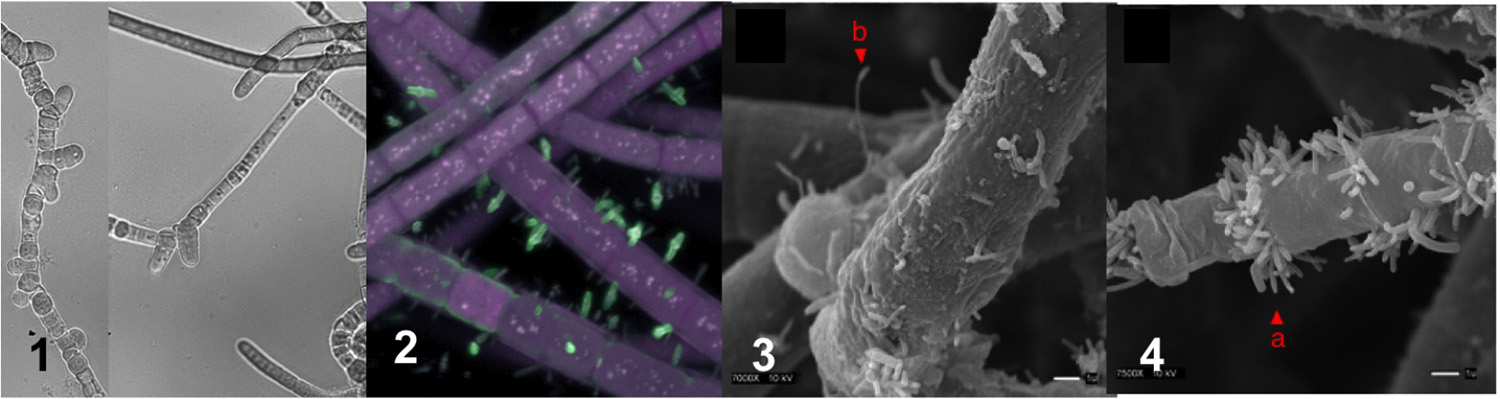
Not only that, using light, fluorescence and scanning electron microscopy, they found that the culture includes many other bacteria, some that were attached to the cyanobacterium’s filaments and some that weren’t attached.
In the AEM paper, the researchers describe how they sequenced the genome of the T. nodosa strain, called HT-58-2, as well as the genome of a second-most abundant bacterial species found in the culture. The cyanobacterium appears unable to grow without other bacteria, forming a type of consortium or symbiosis.
“It’s a complex microbial community,” Miller said. “And that raises the question of which organism – the cyanobacterium or the associated bacteria — actually makes tolyporphin.”
Tolyporphins are in a class of chemical compounds known as tetrapyrroles. Pyrroles are a five-atom ring with four carbon atoms and one nitrogen atom, and tetrapyrroles contain four of these rings. Miller and Lindsey saw scattered throughout the chromosome the biosynthetic machinery that HT-58-2 uses to make tetrapyrroles and tetrapyrrole products like chlorophyll, vitamin B-12 and heme.
They also noted a dense cluster of genes, some of which are for tetrapyrrole biosynthesis and some that are unique to the cluster. “We believe this is the hint – a good suggestion – that this cluster could be for the production of tolyporphin,” Miller says.
Next Steps
[pullquote color=’red’ align=”right”]Photosynthetic microbial systems like cyanobacteria have properties that could be beneficial for pharmaceutical production, biofuels and more.[/pullquote]The scientists are now following up on that hint. They plan to clone the cluster and express it in another bacterium like E. coli, to see if that enables E. coli to produce tolyporphin. If so, they’ll then inactivate those genes to see if the E. coli no longer produces tolyporphin. If it doesn’t, Miller explains, that would prove their hunch.
Most likely the story won’t end there: Miller sees the work as a necessary first step in understanding the biosynthesis and role of tolyporphin in cyanobacteria and how they might be used in medicine or to create other products.
“The more we can learn about this strain the better,” Miller says, “because the photosynthetic microbial systems like cyanobacteria have properties that could be beneficial for pharmaceutical production, biofuels and more. Cyanobacteria are also known to be beneficial for nutrient cycling, plant growth and water quality.”